Advancing Preclinical Science with Integrated Expertise
Advancing Preclinical Science with Integrated Expertise
Endpoint Preclinical is a consulting network for preclinical research, supporting CROs, pharmaceutical, biotech, and medical device teams through coordinated surgical, pathology, toxicology, data analytics, and regulatory expertise. Our distributed model provides full-pipeline support through a single point of contact, integrating study design, production-level surgical execution, quantitative analysis, and safety evaluation across therapeutic and device programs.
In the context of toxicology and safety studies, this structure enables aligned support for PK, PD, and TK programs — including vascular and non-vascular cannulation services delivered in collaboration with PRA — ensuring procedural consistency and downstream integration with pathology, data interpretation, and regulatory documentation.
Explore our expertise in preclinical safety and toxicologic interpretation below.
Safety & Toxicology Experts
Eric Adams, MS, SRS
A preclinical surgical specialist supporting nonclinical safety and toxicology programs, Eric Adams provides consistent model execution aligned with defined safety endpoints. He works with study teams to ensure surgical implementation supports tissue integrity, reliable sample collection, and downstream pathology and toxicologic evaluation.
Joel Baublits, MS
A biostatistics specialist supporting nonclinical safety and toxicology programs, Joel Baublits applies advanced statistical methodologies to integrate multimodal cardiovascular, respiratory, and toxicology datasets. His work aligns data analysis with study design and regulatory expectations, strengthening interpretation of safety pharmacology and preclinical endpoint outcomes.
Brad Bolon, DVM, PhD, DACVP, DABT, FRCPath
Internationally recognized toxicologic pathologist with decades of experience in nonclinical safety assessment and neuropathology. Dr. Bolon provides high-level interpretive consultation and independent pathology peer review for complex or high-stakes programs where clear conclusions are essential to regulatory and development decisions.
Ryan Boyd, DVM, MS, DACVO
A board-certified veterinary ophthalmologist, Ryan Boyd supports nonclinical safety and translational programs involving ocular therapeutics, gene therapy, and intraocular device evaluation. His expertise includes subretinal and intravitreal delivery, advanced ocular imaging, and GLP-aligned study execution. Ryan also supports toxicological eye examinations in preclinical safety studies, contributing specialized ophthalmic assessment to support reliable safety characterization and regulatory documentation.
Ray Chui, PhD, DSP
A specialist in cardiovascular and respiratory data analytics, Ray Chui supports nonclinical safety pharmacology and toxicology programs through rigorous analysis of hemodynamic, ECG, and respiratory endpoints in GLP and non-GLP studies. He develops end-to-end data interpretation and reporting strategies aligned with regulatory standards, ensuring cardiovascular and respiratory safety data are accurately characterized and clearly documented within preclinical programs.
Jessica Durrant, DVM, DACVP
Board-certified veterinary pathologist and Director of Pathology at DTR Labs, specializing in quantitative and digital pathology workflows supporting efficacy, biodistribution, and nonclinical safety studies. Dr. Durrant is particularly well suited for early-phase and translational programs requiring quantitative pathology and thoughtful alignment between study objectives and downstream tissue analyses.
David W. Eaker, PhD, DABT
A board-certified toxicologist, David Eaker supports nonclinical safety programs through preclinical strategy, toxicologic risk assessment, and regulatory consulting. He works with study teams to align toxicology, biocompatibility, and analytical data with regulatory expectations, strengthening the interpretation and documentation of safety findings.
Brad Gien, BSc, CM
President of Preclinical Research Associates (PRA), Brad Gien supports nonclinical safety programs through scalable, production-level surgical execution delivered within client laboratories. His work aligns surgical model implementation with GLP and IND-enabling studies, ensuring procedural consistency supports downstream pathology, toxicology, and regulatory evaluation.
Tim Hacker, PhD
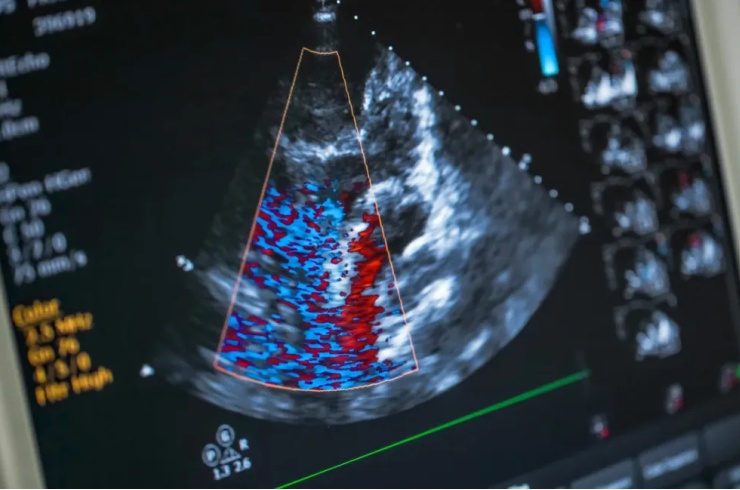
A cardiovascular research specialist, Tim Hacker supports nonclinical safety programs through advanced sonography, surgical model execution, and hemodynamic data analysis. He leads Endpoint’s rodent echocardiography services and provides expertise in ventricular PV-loop assessment aligned with defined cardiovascular endpoints. His work integrates functional cardiac data into toxicology and safety pharmacology evaluations.
Stacy Hosking, DVM, DACVIM (Cardiology)
A board-certified veterinary cardiologist, Stacy Hosking supports nonclinical safety pharmacology and toxicology programs through advanced echocardiography and ECG analysis. She provides on-site and remote cardiac imaging services aligned with study design and defined cardiovascular endpoints. Her work integrates imaging and electrophysiologic data into safety assessments and regulatory documentation for therapeutic and device development programs.
Allison Rodgers, BA
A specialist in preclinical ultrasound imaging and data analysis, Allison Rodgers supports cardiovascular and pulmonary safety programs through high-quality image acquisition and structured data reporting. Her expertise includes optimization of echo protocols aligned with defined functional endpoints. Allison’s work ensures consistent imaging data that integrate effectively into toxicology and safety pharmacology evaluations.
Resource Library
Access all of the digital materials we are sharing at SOT and ToxExpo 2026.

How to Get Started
1. Discovery Meeting
Meet with our leadership team to discuss your research goals, challenges, and timelines. We’ll identify the right experts within our network to support your studies, whether you need specialized guidance, training, or full study coordination. A CDA can be enacted at any time to maintain confidentiality.
2. Sign an Agreement
Endpoint’s streamlined contracting process removes administrative friction. One master service agreement allows your organization to quickly access multiple consultants, capabilities, and study types without negotiating terms for every engagement.
3. Start Placing Our Experts
Once your MSA is in place, Endpoint experts can be integrated into ongoing or upcoming research programs. From study planning and design to regulatory support, our network enables teams to expand preclinical capacity as program needs evolve.