How to Plan IND-Enabling Ocular Studies: Dose Route and Immune Response
How to Plan IND-Enabling Ocular Studies: Dose Route and Immune Response
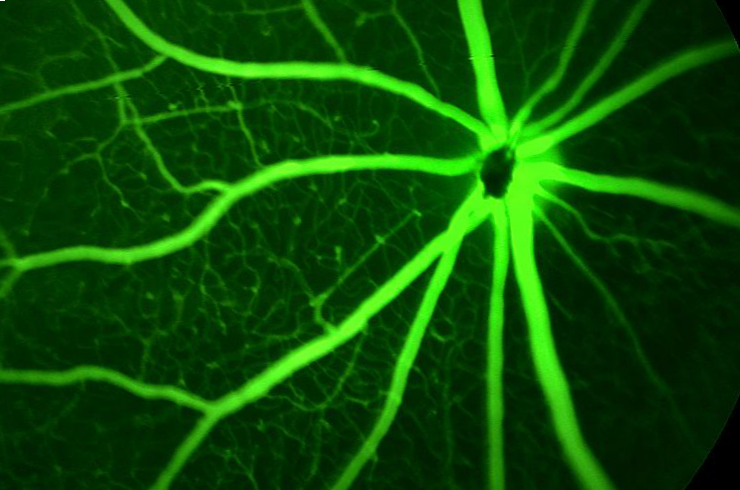
IND-enabling ocular studies are shaped by study design, dose route and species selection. This webinar focuses on how to plan and interpret them effectively, and how dose route selection impacts the ocular immune response.
Ocular preclinical studies require careful consideration of how study design choices—particularly administration strategy—will influence both outcomes and interpretation. While the eye is often described as an immune-privileged site, intraocular administration can still elicit a range of innate and adaptive immune responses that complicate the evaluation of safety and pharmacologic effect.
This webinar will examine how to approach these challenges when planning IND-enabling programs. Drawing on practical examples, Dr. Ryan Boyd will discuss how immune responses arise in preclinical ocular studies, how dose route impacts their development and progression, and how they can impact regulatory interpretation. The focus will be on identifying potential confounding factors early and making study design decisions that support clear, interpretable data.
Key Learning Objectives:
- Understand how immune privilege in the eye influences, but does not eliminate, immune responses following intraocular administration
- Identify common sources of immune-related findings in ocular preclinical studies, including procedure-related effects, formulation factors, and therapeutic-specific responses
- Distinguish between innate and adaptive immune responses and how they evolve over time in preclinical studies
- Evaluate how study design influences translational relevance and regulatory interpretation
- Apply practical considerations to plan IND-enabling ocular studies that support clear, interpretable outcomes
Speaker
Ryan Boyd, DVM, MS, DACVO
Expert in Preclinical Ophthalmology, Gene Therapy, and Surgical Techniques
Dr. Ryan Boyd is a board-certified veterinary ophthalmologist with extensive expertise in preclinical ophthalmology, advanced surgical techniques, and gene therapy for retinal diseases. With a decade of full-time experience in GLP-compliant research environments, Ryan specializes in subretinal, suprachoroidal, and intravitreal injections, intraocular device evaluation, and ocular imaging. His work supports the development of therapeutics and medical devices for preclinical models, helping clients achieve reliable and reproducible results.